Is Melting Exothermic Or Endothermic
Endothermic vs. Exothermic with Examples
Chemic reactions are classified as endothermic or exothermic based on the energy transfer between the surrounding environment and the system where the reaction is taking place. There are two methods for determining whether a chemical reaction is endothermic or exothermic:
a. find the temperature modify or the rection
b. Determine the enthalpy alter (ΔH) between the reactants and the products.
The primary stardom betwixt endothermic and exothermic reactions is that endothermic reactions absorb free energy from their surround, whereas exothermic reactions release energy into them. In addition, A chemical reaction with a positive ΔH is said to be endothermic, while a chemical reaction with a negative ΔH is said to be exothermic.
What is an Exothermic Reaction?
The temperature of the products in endothermic reactions is typically lower than that of the reactants as shown in the figue beneath.
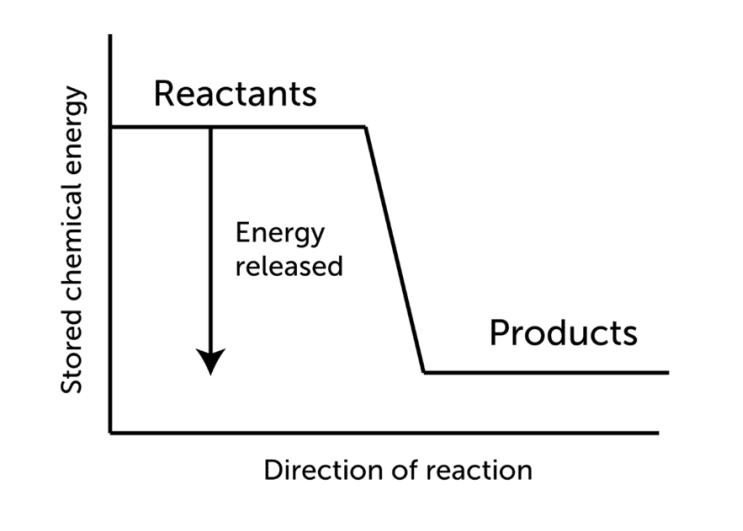
Exothermic reactions crave additional energy to catechumen reactants into products. As a effect, for exothermic reactions, the modify in enthalpy, denoted past delta ΔH, will be negative.
Examples of Exothermic Reactions
- When h2o freezes into ice cubes, the energy is released in the form of heat.
- The process of snow formation is an exothermic reaction
- Setting Cement And Concrete
- Combustion process
- Rusting of fe
- lighting a candle is an exothermic reaction
- phase transitions from the liquid to solid land
- Digestion of food
- the reaction between quick lime and h2o is exothermic
- freezing of water in an ice is an exothermic reaction
- Sponification procedure
- Thermite welding
What is an Endothermic Reaction?
The temperature of the products in exoothermic reactions is typically in a higher place than that of the reactants as shown in the figue below.
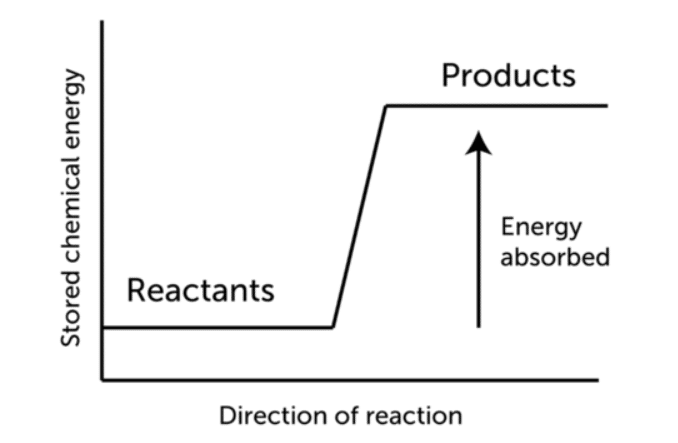
Enthalpy is absorbed from the environs as reactants are converted to products in an endothermic reaction. The reaction's enthalpy change is positive. For instance, In the instance of methane combustion, the enthalpy alter (ΔH) is negative because heat is released by the arrangement.
Examples of Endothermic Reactions
- Sweating is an endothermic process
- Boiling of Water
- photosynthesis
- evaporating liquids
- alkane cracking
- Cooking due south an endothermic reaction
- Melting is an endothermic procedure
- dissolving tabular array table salt in water is endothermic
- vaporization (liquid to gas)
- Condensation reaction
- Sublimation procedure
Departure between Exothermic and Endothermic Reactions
| Endothermic | Exothermic |
| A reaction in which the system absorbs free energy in the form of heat from its surroundings. | A reaction that releases heat as a type of free energy from the organisation. |
| The energy from the environment is absorbed into the reaction. | The system's energy is released into the environment. |
| Oestrus is released as a kind of energy. | Heat, electricity, light, and sound are released every bit a kind of energy. |
| Examples include ice melting, evaporation, cooking, gas molecules, and photosynthesis. | Examples include rusting atomic number 26, settling, chemical bonds, explosions, and nuclear fission. |
| entropy increases (ΔS > 0) | disorder decreases (ΔS < 0) |
| increase in enthalpy (+ΔH) | subtract in enthalpy (-ΔH) |
| Examples of an endothermic reaction: – Photosynthesis – Melting ice – Evaporating liquid h2o – Not bad alkanes – Cooking an egg | Examples of an exothermic reaction: – Combustion reaction – Respiration – Corrosion of metal (an oxidation reaction) – Dissolving an acrid in water |
Summary
- Energy is constantly released during an exothermic reaction, often in the form of heat.
- Exothermic reactions characterise all combustion reactions. A substance burns when it combines with oxygen, releasing energy in the form of heat and light.
- Endothermic reactions require a constant input of energy, often in the form of heat.
- Photosynthesis is i of the most important endothermic reactions. Low-cal provides the energy required for photosynthesis.
Frequently Asked Questions
ane. How combustion is an exothermic reaction?
All combustion reactions are exothermic. A substance burns when information technology combines with oxygen during a combustion reaction. When substances fire, they usually emit energy in the grade of rut and light.
For case, The combustion of wood is an exothermic reaction that releases a lot of free energy as oestrus and calorie-free.
2. Exercise exothermic reactions increase entropy?
The external entropy (entropy of the surroundings) increases during an exothermic reaction. The external entropy (entropy of the environment) decreases during an endothermic reaction.
More Links
Is Melting Exothermic Or Endothermic,
Source: https://whatsinsight.org/endothermic-vs-exothermic/
Posted by: phillipscoully.blogspot.com


0 Response to "Is Melting Exothermic Or Endothermic"
Post a Comment